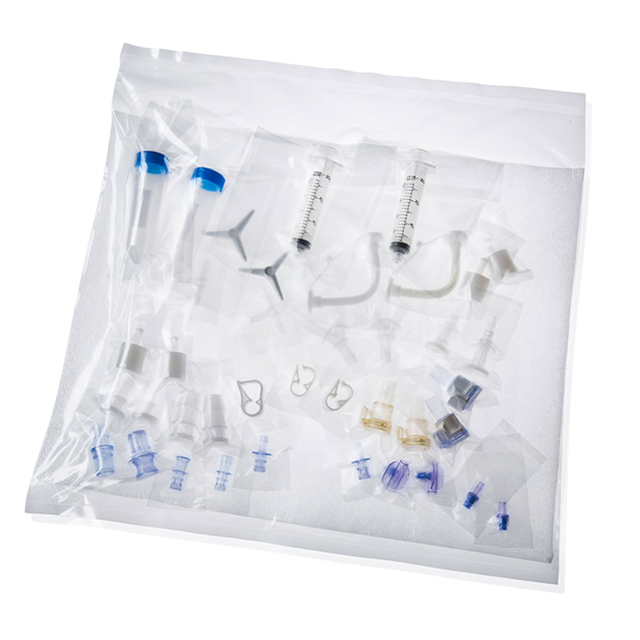
Sterile packaging, offering enhanced flexibility
Components can be packaged in one single kit, designed for ease of use for the end user - allowing for the maintenance of sterile components while opening the bag under a hood. By eliminating the need for on-site sterilization, components can enhance efficiency and provide greater flexibility when working with multiple components or when requiring sterility over numerous uses.
Process and sterilization
- Available for any quantity of parts
- Flexibility to your process
- Ability to be completed for any part or product dependent on chemical compatibility for the proper sterilization method (Gamma, Autoclave, and EtO Sterilization)
- Innovative process development kits
- Comprehensive compatibility guide
Product and packaging features
- Individually double-bagged
- Products can maintain ‘sterile’ status even after the initial packaging is opened due to the bagging process
- See-through, puncture-proof bags
Bioprocessing sterility, from start to finish
Our Sterile Individually Packaged Components can be used in all areas of bioprocessing, from research and development to fill-finish.