Results 1 - 10 of 20
 Article
Article
Optimized Handling of DPTE-BetaBag® Tyvek in steam sterilization
Following specified load and autoclave cycle parameters will result in the best performance for DPTE-BetaBag® Tyvek®. These easy steps will improve outcomes and reduce the risk of bag…
View Article
Article
Top 3 Causes of Cross Contamination in Biosafety Facilities
Cross contamination in BSL-3 and BSL-4 laboratories represents a significant risk to personnel and the surrounding community due to the dangerous agents housed within these facilities.
View Article
Article
5 Factors for Safe Material Transfer in BSL-3 and BSL-4 Facilities
BSL-3 and BSL-4 laboratories highlight the fact that if proper protocols are not followed, contamination can cause detrimental results.
View Product
Product
GSS P Steam Sterilizer
This Sterilizer sets a new standard for reliable contamination prevention in biopharmaceutical production. GSS P is a steam sterilizer from Getinge for component sterilization in…
View Product
Product
Getinge Lancer LSS Laboratory Steam Sterilizers
Getinge Lancer LSS Laboratory steam sterilizers offer high-performance autoclaving of labware, process materials, and byproducts used in laboratory applications.
View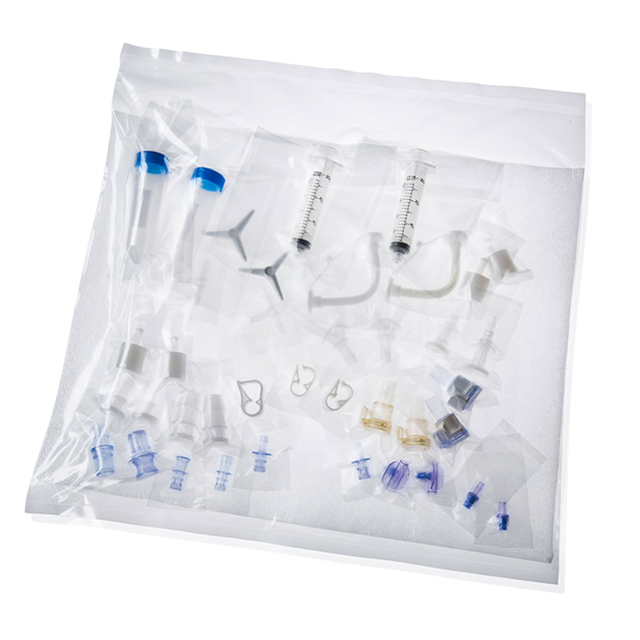 Product
Product
Packaged Components
Sterile individually packaged components The Sterile individually packaged components offer a convenient and flexible solution for your sterile needs, featuring double-bagged components…
View Product
Product
GSS L & R Steam Sterilizer
GSS L & R helps you achieve predictability, reliable containment and scientific data integrity in biomedical research facilities. Sterilization, decontamination and biocontainment…
View Solutions
Solutions
Biocontainment in Vaccine Production
All waste material generated must be considered bio-hazardous and must be treated with care before disposal. This means safe handling and effective bio-decontamination.
View Product
Product
HPConnexx
Custom single-use assemblies HPConnexx™ offers comprehensive and customizable assemblies to support every stage of your bioprocess, from research and development to large-scale…
View


