See and deliver what your patient wants
In most intensive care units, 20% of the patients consume 80% of ventilation resources, which may lead to increased complications and unwanted outcomes. [31] For these patients, conventional ventilation simply isn’t enough. By using the NAVA mode on Servo ventilators, which works regardless of patient category or size, the ventilator shows you what the patient wants. Together with other personalized ventilation tools, this can help you reduced complications [9] [10] [29] [30], monitor and reduce sedation [5] [19] [20] [21], achieve earlier and more successful weaning [3] [4] [8] [13] [14] and shorten the time of mechanical ventilation [3] [20] [21].
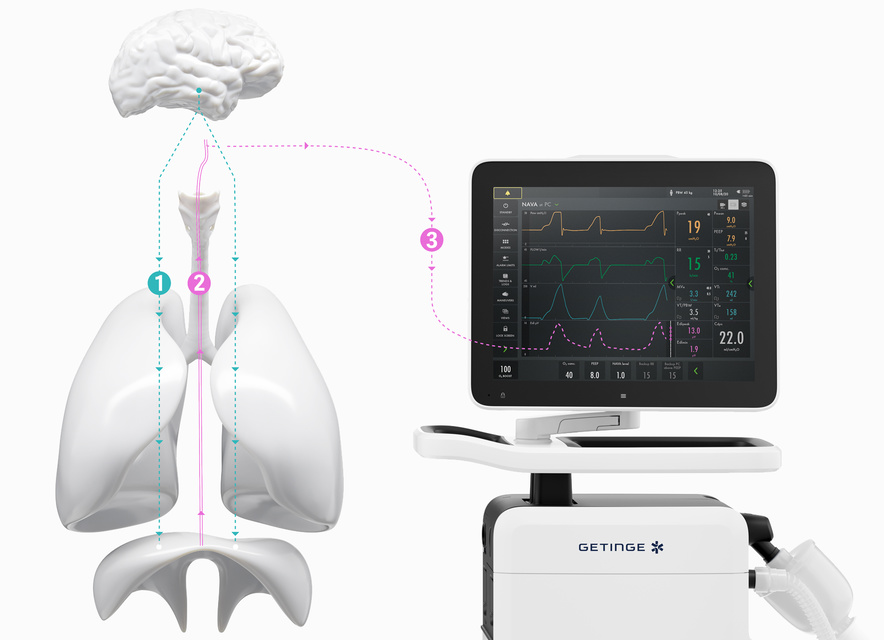
How NAVA delivers a more personalized level of ventilation in three easy steps.
How NAVA delivers a more personalized level of ventilation in three easy steps.
The brain directs our breathing.
The size and timing of each breath is controlled by the brain’s respiratory center. When the brain has processed multiple sensory inputs, it sends a signal via the phrenic nerve that electrically activates the diaphragm, leading to muscle contraction. The diaphragm then contracts into the abdominal cavity, leading to a descending movement, creating chest wall and lung expansion and inflow of air.
Micro-electrodes detect the vital sign of respiration (Edi).
The electrical discharge of the diaphragm is captured by a special nasogastric feeding tube fitted with an array of electrodes (the Edi catheter). Like an ordinary enteral feeding tube, the Edi catheter passes through the esophagus, where it measures the electrical activity of the diaphragm. The electrodes also monitor ECG signals, which are used to verify the position of the Edi catheter.
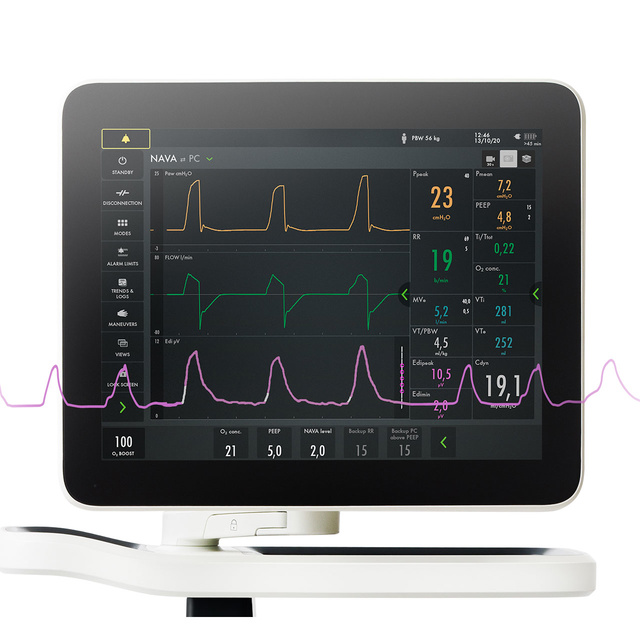
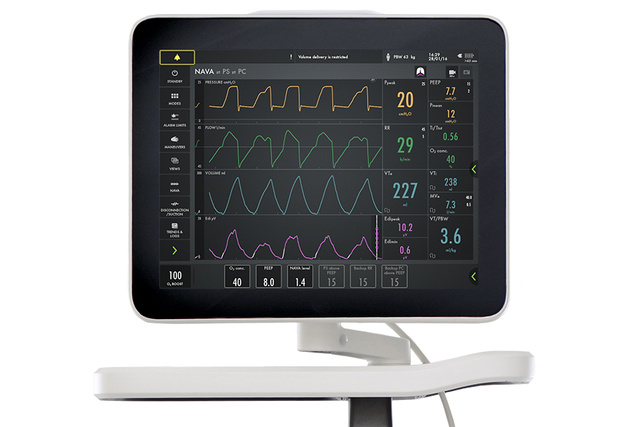
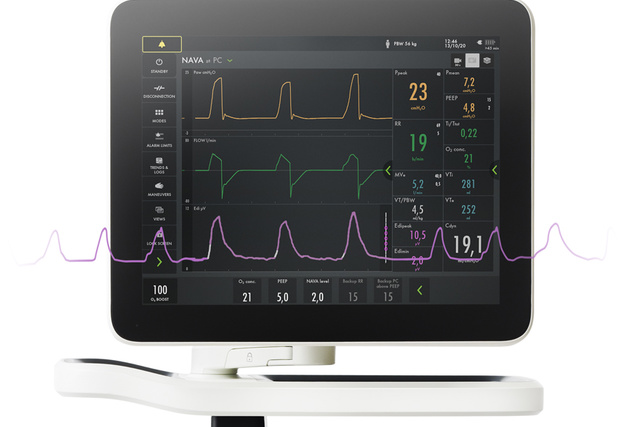
Your Servo ventilator user interface presents it all.
The lower part of the screen shows the Edi signal at all times, even if the patient ventilation mode is not switched to NAVA. When you switch on the NAVA mode, the Edi signal automatically guides the ventilator to deliver support in time with and proportion to the diaphragm. The ventilator acts as a second inspiratory muscle – in perfect synchrony with the patients.
The benefits of Edi
The diaphragm is the "heart" of the respiratory system and is designed to be continuously active. [26] The Edi is a bedside diagnostic tool that allows you to monitor and safeguard the patients diaphragm activity. [27] [28] The Edi guides weaning [29] and helps you prevent muscular exhaustion during weaning trials, even after extubation. [30]
Achieve faster personalized weaning with lung- and diaphragm-protective ventilation
1. Monitor Edi – the vital sign of respiration, from day zero
2. Protect and activate the diaphragm to wean earlier
3. Protect the lungs in synchrony with the patient

4. Improve the patient’s overall ICU experience
Monitor Edi – the vital sign of respiration
In addition to monitoring the impact of mechanical ventilation on lung function, it is also imperative to monitor patient respiratory drive and effort from day zero. Edi monitoring facilitates earlier and more informed decision-making. With this vital sign in your lower screen, you can detect diaphragm inactivity, over-sedation, patient-ventilator asynchrony as well as over- and under-assist. You can also monitor increased work-of-breathing during weaning trials and post-extubation[5] [6] [7] [8] [9] [10] [11] [12] [13] [14] [15].

Diaphragm-protective ventilation
Suboptimal mechanical ventilation can rapidly cause acute diaphragm atrophy or load-induced injury associated with poor clinical outcomes [16] [17]. The key physiological benefits of NAVA are that pressure is always delivered in proportion to and in synchrony with the patient’s own respiratory drive, and that Edi is readily available as a bedside diagnostic tool [1] [2]. NAVA shortens the duration of weaning and increases the proportion of patients with successful weaning[3][4].

Lung-protective ventilation
A key difference between NAVA and conventional support modes is that tidal volume (VT) is controlled via neuro-electrical output from the patient’s respiratory center. Lung over-distension is thus prevented thanks to the Hering-Breuer reflex, which down-regulates the respiratory drive at higher tidal volumes to avoid hyperinflation. As a result, it is possible to achieve lung-protective spontaneous breathing within the protective range of 6-8 ml/kg [1] [2] [18] PBW.
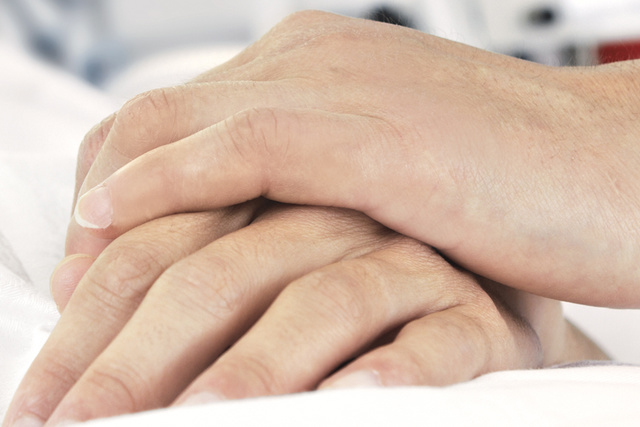
Improved patient experience
NAVA has been shown to improve the patient’s overall ICU experience, helping clinicians to potentially reduce sedation with improved comfort and sleep quality [19] [20] [21] [22] [23]. Together, Edi and NAVA assure that the breathing efforts from all patient categories are effectively assessed and responded to. For patients with acute exacerbation of COPD, our non-invasive, leakage-independent NIV NAVA mode can be helpful effective in managing their status and avoiding intubation [14] [24] [25] [26] [27] [28].
Personalized support throughout the treatment
Invasive NAVA
Synchronized assist, weaning and sedation management, supporting early diaphragm activation.

Non-invasive NAVA
Synchronized assist, independent of leakages allowing a gentler mask application.
Edi monitoring
Monitor diaphragm activity and breathing effort after extubation. Can be used with High Flow therapy if needed.
Marketing Sales - Brochures
-
Servo-u gives you many options for personalized lung protection and weaning, for treatment of all patient categories. All are easy to understand, implement and use.
-
Servo-n offers an advanced all-in-one solution for personalized lung protection and weaning, helping neonates breathe, sleep and grow.
-
Achieve faster personalized weaning with lung and diaphragm-protective ventilation.
-
Diaphragm monitoring, helping you improve mechanical ventilation
Marketing Sales - Data Sheet
-
Detect electrical activity of diaphragm, with our range of Edi catheters, enabling NAVA and NIV NAVA ventilation modes, and available in all patient categories.
Marketing Sales - Pocket Guides
-
Edi catheter - with ENFit connector pocket guide
Clinical - Articles
-
Clinical literature on NAVA, NIV NAVA, and Edi monitoring demonstrating the benefits in adult patients, such as improved synchrony, comfort, and tidal volume variability. Studies highlight their effectiveness in promoting weaning, extubation, and supporting ECMO and ECCO₂R.
-
Clinical literature on NAVA, NIV NAVA, and Edi monitoring demonstrating the benefits in neonatal and pediatric patients, such as improved synchrony, comfort, and oxygenation. Studies highlight their effectiveness in promoting weaning, extubation, and reducing sedation needs.
Training Material
-
Flowchart for adult patients, covering NAVA terminology, Edi catheter insertion and positioning, NAVA set-up, workflow, weaning in NAVA and troubleshooting.
-
Flowchart for neonates, including NAVA terminology, Edi catheter insertion and positioning, NAVA set-up, weaning in NAVA and troubleshooting.
Visit our Academy – training and education designed to enhance your proficiency
For more information about our onsite events or remote trainings, you can also contact your local sales & service representative.