Resultat 61 - 66 av 66
 Dokument
Dokument
FEB2021 475699 Ripple 20
Datascope/Getinge is initiating a voluntary Medical Device Correction for the Cardiosave Hybrid and Cardiosave Rescue Intra-Aortic Balloon Pump (IABP) due to cybersecurity vulnerabilities…
View Dokument
Dokument
TUV Certificate No. QS2 102541 0002 Rev. 04
TUV Certificate No.QS2 102541 0002 Rev. 04 / ISO 13485:2016 IABP and Accessories, HL40, Collagen Effective Date 2023-02-06 Exp. Date 2026-02-05
View Dokument
Dokument
JUNE2023 848201 11 Poor or No ECG Signal_Docking Power Battery Failure
Datascope Corp., a subsidiary of Getinge is initiating a voluntary Medical Device Correction for the Cardiosave Hybrid and Cardiosave Rescue Intra-Aortic Balloon Pump (IABP) due to the…
View Dokument
Dokument
SEPT2021 527856 Li-Ion Battery Packs
Datascope/Getinge is initiating a voluntary Medical Device Removal for a limited number of Cardiosave Li-Ion Battery Packs with Part Number/REF Number 0146-00-0097 used with Cardiosave…
View Sida
Sida
Product Catalog
Explore Getinge's comprehensive range of advanced healthcare and life sciences solutions.
View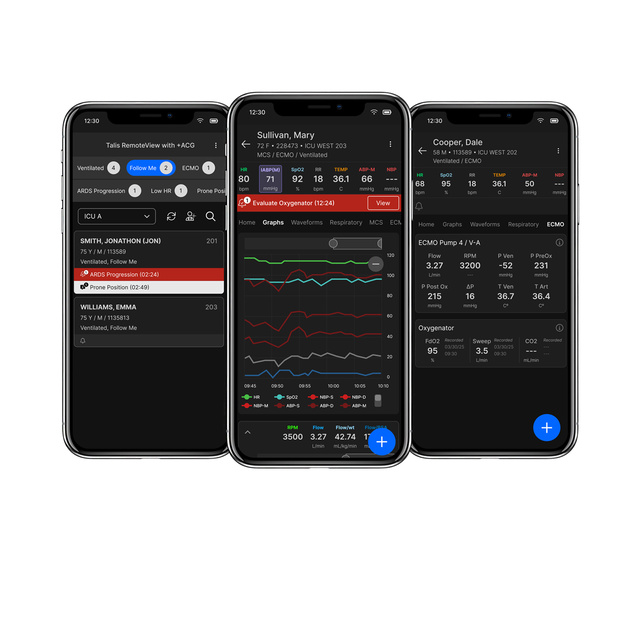 Produkt
Produkt
Talis RemoteView with +ACG
Clinical management and surveillance system with configurable clinical decision support Talis RemoteView with +ACG is an advanced platform for high-acuity care, empowering clinicians to…
View

